What if we made photosynthetic mice?
A brilliant and bizarre way to tackle degenerative diseases
This study investigates the use of nanosized photosynthetic units derived from plants to improve intracellular anabolism in pathological conditions. The units, called nanothylakoid units (NTUs), are encapsulated in a specific cell membrane to enable cross-species applications. When exposed to light, the NTUs increase levels of ATP and NADPH in cells, improving anabolism and restoring cellular metabolism and protecting against degenerative diseases like osteoarthritis.
Welcome to the 44 new Evolutionaries who have joined us since last month! If you haven’t subscribed, join 3,000+ smart, curious folks by subscribing here:
Sean’s Summary of:
A plant-derived natural photosynthetic system for improving cell anabolism
Background
Sometimes, when people are sick or hurt, their bodies don't have enough energy or substances that help with building new cells. There is a process in the body that helps make energy, called the tricarboxylic acid (TCA) cycle, but sometimes trying to directly fix this process can cause problems with how the body works.
Using natural systems to make energy and substances that help with building new cells can create new opportunities for treatment. Plants use a system that takes sunlight and transforms it into an energy that cells can use (ATP and NADPH). When trying to use this approach to treat people, it can be difficult for the body to accept the treatment and not reject it.
The cell membrane is important for keeping the inside of a cell protected. Scientists can use small parts of the cell membrane called vesicles to put special materials inside to help them work better in the body. These vesicles can be used to help reduce the possibility of the body rejecting these energy producing systems.
Osteoarthritis is a common degenerative disease. Chondrocytes with an energy deficit exhibit decreased synthesis of extracellular matrix (ECM) proteins. Current treatments cannot systematically correct the metabolic imbalance. If an energy producing system could be transplanted into chondrocytes, would it be enough to fix the disease?
Hypothesis
If a plant-derived photosynthetic system could be accepted by an animal cell, could it then be used to enhance anabolism in those cells and make up for energy deficits that lead to disease?
Results
The nanothylakoid units (NTUs) were successfully developed and encapsulated in a specific type of cell membrane, the chondrocyte membrane (CM), to enable cross-species applications.
The CM-NTUs were able to enter cells, avoid degradation, and increase intracellular levels of ATP and NADPH following exposure to light.
The increases in ATP and NADPH levels improved anabolism in degenerated cells and may have potential as a therapeutic strategy for degenerative diseases.
The CM-NTUs may be able to restore cellular metabolism and improve cartilage homeostasis, and may protect against the progression of osteoarthritis.
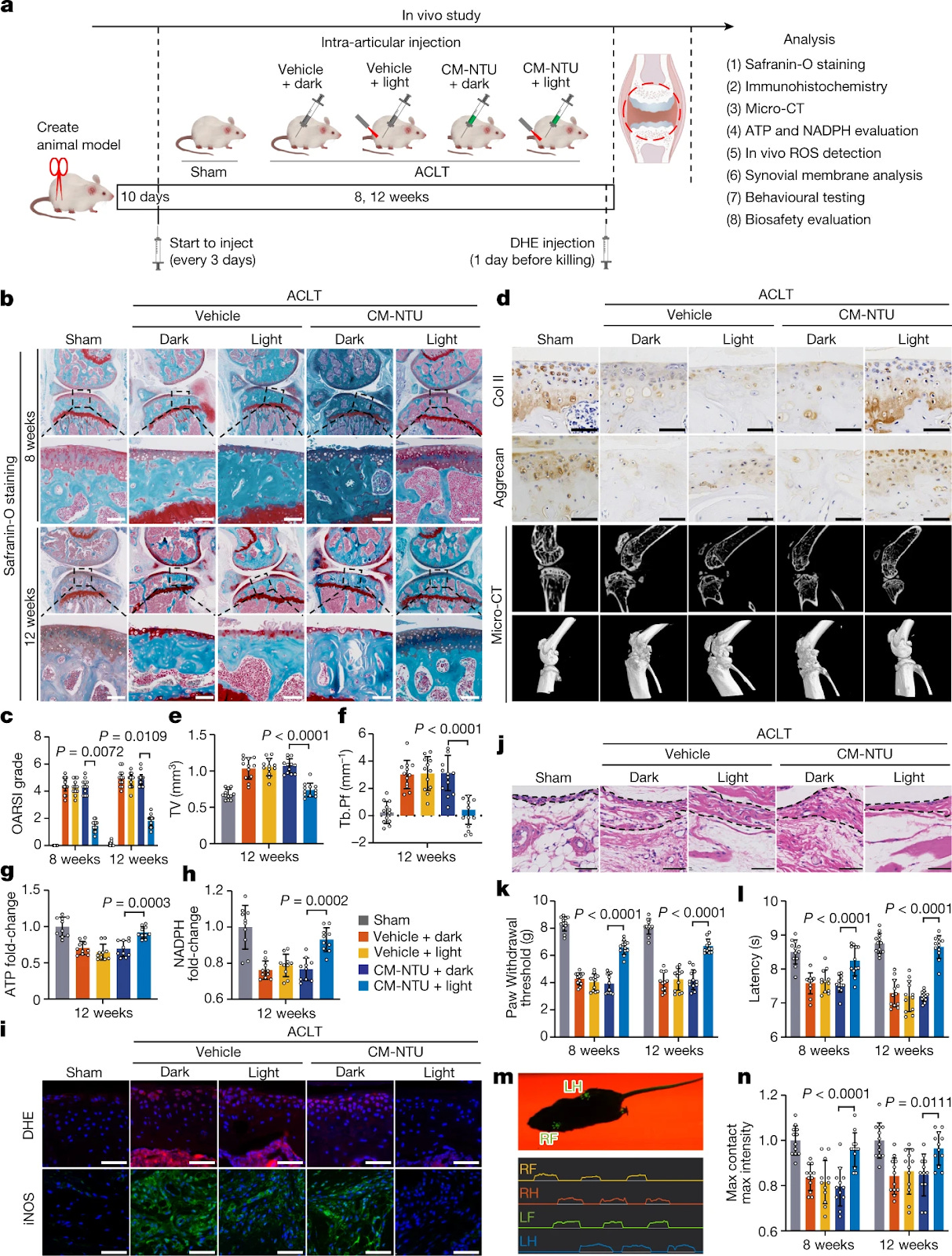
Fig. 5: In vivo effect of CM-NTU treatment on osteoarthritis in mice. The in vivo effect of CM-NTU treatment on osteoarthritis was investigated in 12-week-old male mice. a, Schematic illustration of establishment of the mouse model of osteoarthritis and the experimental design to evaluate the protective effects of CM-NTUs. DHE, dihydroethidium. b, Safranin-O staining of joint sections at 8 and 12 weeks. c, Medial tibial plateau joint score based on the OARSI scoring system (n = 12, mean ± 95% confidence interval (CI)). d, Immunohistochemical staining (Col II and aggrecan) of joint sections at 12 weeks (top two rows), sagittal views of micro-CT images of the knee joints (third row) and three-dimensional images of the knee joints at 12 weeks (bottom row). e,f, Twelve weeks after the operation, quantitative analysis of total tissue volume (TV) (e) and trabecular pattern factor (Tb.Pf) (f) in subchondral bone (n = 12, mean ± s.d.) in mice. g,h, ATP (g) and NADPH (h) levels in CM-NTU-treated joints at 12 weeks (n = 10, mean ± s.d.). i, ROS fluorescence and immunofluorescence of iNOS in CM-NTU-treated joints at 12 weeks. j, H&E staining of synovial membranes in CM-NTU-treated joints at 12 weeks. k,l, Electronic von Frey (k) and hotplate (l) pain assays in mice at 8 and 12 weeks after the ACLT operation (n = 12, mean ± s.d.). m, Schematic of gait analysis. RF, right front; RH, right hind; LF, left front; LH, left hind. n, Gait assessment scores for maximum contact maximum intensity (right hind limb) in mice 8 and 12 weeks after operation (n = 12, mean ± s.d.). n represents the number of mice per group. P values are shown in graphs and were determined using nonparametric Kruskal–Wallis test (c) or one-way ANOVA (e–h,k,l,n). Scale bar, 50 μm (d,i,j) or 100 μm (b).
Limitations
The authors didn’t offer up their own limitations in this paper, which I never really like. Experiments almost always have some sort of limitation. I am not so educated in this specific field of plant-derived photosynthetic systems that I can offer limitations of the study itself. Hell, it could be a perfect study. Their analysis checks out, the results speak for themselves very nicely and I love how the actual paper was written in a way that flows through each step of the project. I can see where they’ll branch off to many other projects from this one too, although the authors don’t mention “next step” comments either; also odd. I would say that the limit that comes to my mind is the applicability as the animal model scales up, especially to human size. Mice, especially lab mice, are easy to shine lights though. They’re already pretty transparent. I’d be interested to see this used in non-human primates or dogs, hell even horses (there would be a lot of money in race horse regeneration). I don’t see that as a huge limitation, but one worth noting.